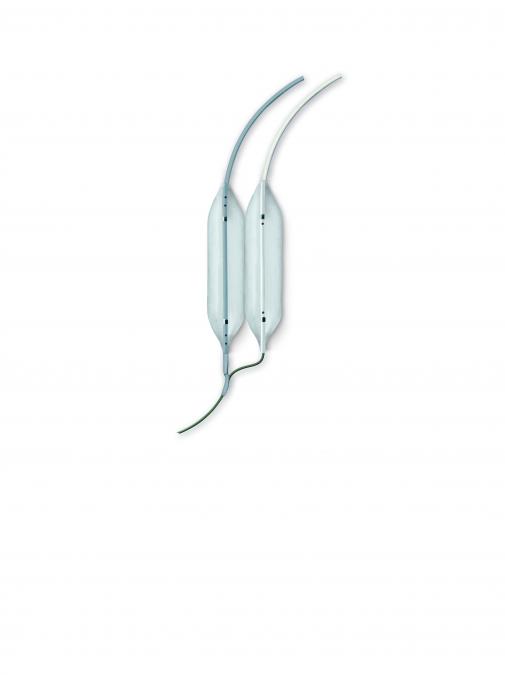
Mitral Dilatation Kit
Kit Components
The kit contains a Multi-Track balloon catheter, a matched rapid exchange balloon catheter, a Multi-Track™ angiographic catheter, a dilator and a super stiff guidewire.
The catheter bodies are DEHP-free and not made with natural rubber latex. The balloons are a thermoplastic elastomer; non-compliant - which will not exceed +/- 10% of the labeled balloon size at RBP, are DEHP-free, and not made with natural rubber latex.
Specifications, Instructions for Use, and Forms
Mitral Kit Components include:
- Multi-Track balloon dilatation catheter (14, 16, 18 or 20mm in diameter) x 5cm in length
- Matched rapid exchange balloon dilatation catheter
- 14 FR, 70cm dilator
- Multi-Track™ angiographic catheter (5 FR x 80 cm)
- Super stiff, 0.035” guidewire with floppy tip
Indication: Percutaneous transvenous mitral commissurotomy in patients with hemodynamically significant mitral valvular stenosis
Mitral Dilatation Kit IFUs:
INTERNATIONAL: RM0344-18i
| Balloon Diameter (mm) | Balloon Length (cm) | Introducer (FR) | Shaft Size (FR) | Usable Length (cm) | Guide Wire (inches) | Rated Burst Pressure (ATM) | Maximum Volume (cc) | Catalog Number | Product |
|---|---|---|---|---|---|---|---|---|---|
| 0.0 | 0.00 | 0 | 0.0 | 0 | 0.000 | 0.0 | BMK14 | MITRAL KIT | |
| 0.0 | 0.00 | 0 | 0.0 | 0 | 0.000 | 0.0 | BMK16 | MITRAL KIT | |
| 0.0 | 0.00 | 0 | 0.0 | 0 | 0.000 | 0.0 | BMK18 | MITRAL KIT | |
| 0.0 | 0.00 | 0 | 0.0 | 0 | 0.000 | 0.0 | BMK20 | MITRAL KIT |
Disclaimer
All products are subject to individual country regulations in regards to the importation and/or sale of these products. Refer to Instructions for Use for a complete listing of indications, contraindications, warnings, and precautions.